


 Aert Medical
Aert Medical










| Still deciding? Get samples of $ ! US$ 5/Piece Request Sample |
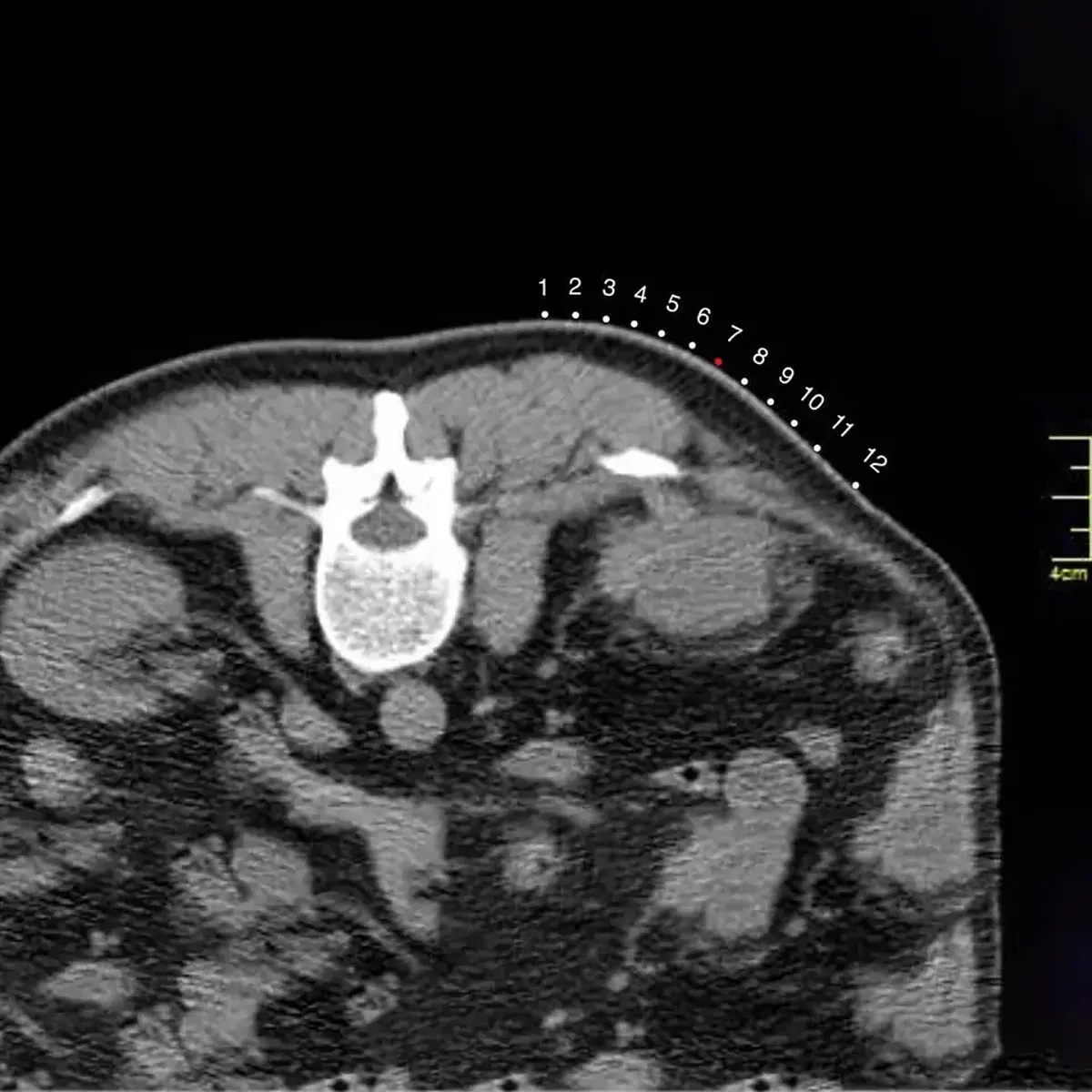
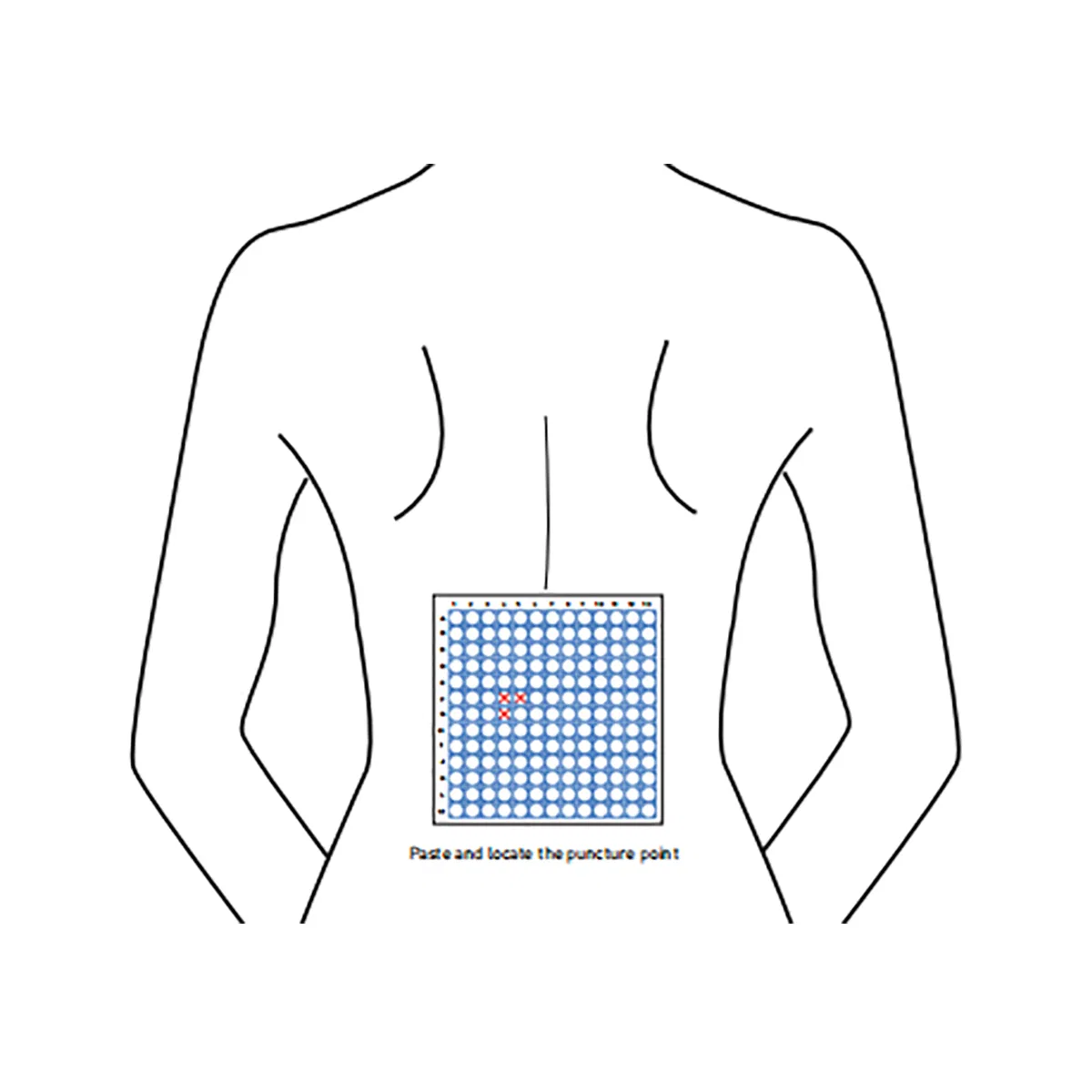
The CT Biopsy Grid is a disposable radiopaque localization grid designed to improve accuracy in image-guided biopsy and drainage procedures. Compatible with CT, DR, DSA, R/F, Ultrasound, and MRI systems, it enables precise alignment between skin markings and imaging coordinates, helping reduce positioning errors, repeated needle adjustments, unnecessary rescans, and patient discomfort.
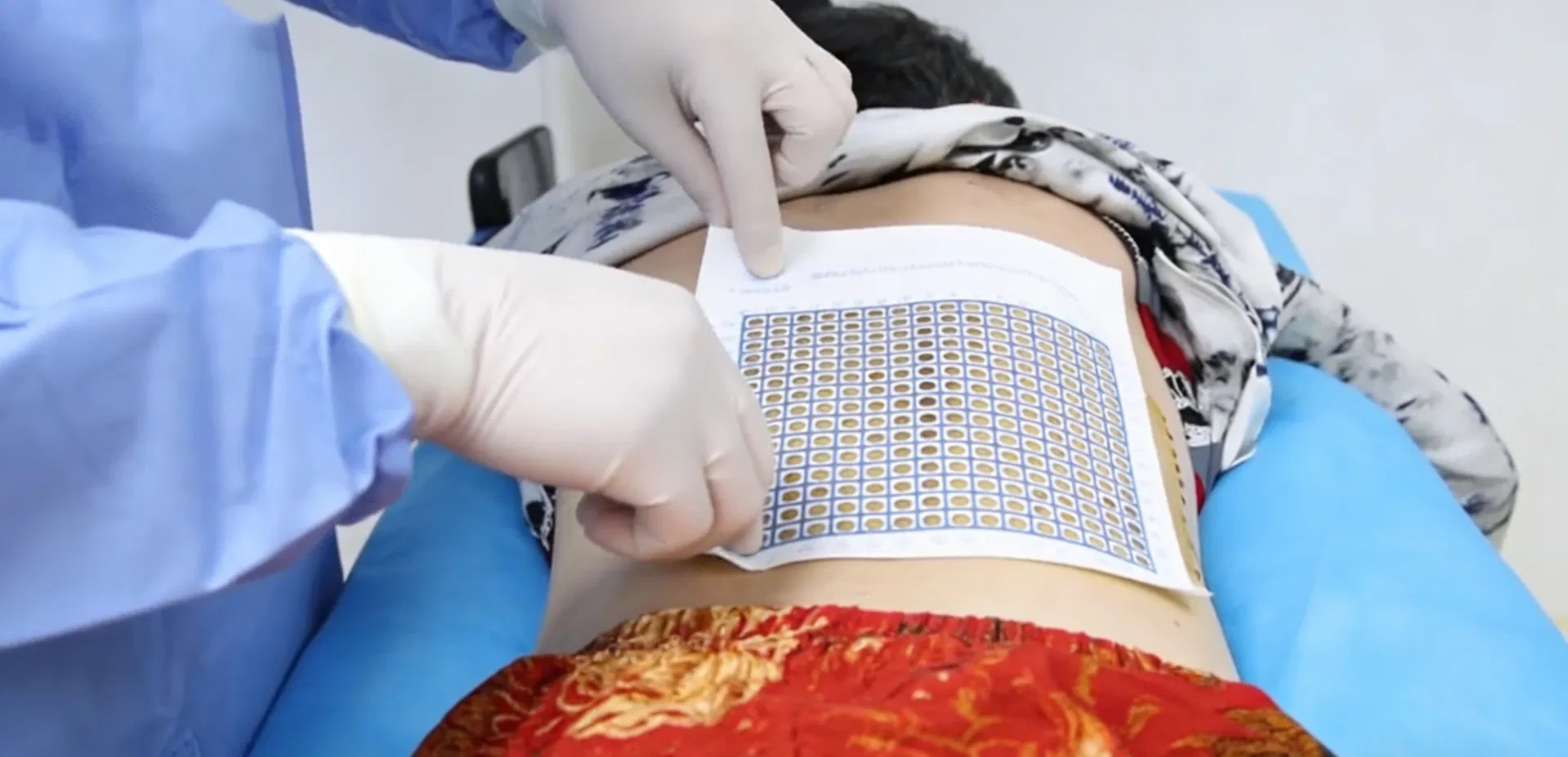
Featuring a full-surface medical-grade adhesive backing, the grid conforms naturally to different body contours and remains flat and stable on the skin throughout imaging. Constructed with a proprietary three-layer design, it includes a lead-free radiopaque ink layer, a non-woven 100% medical cotton pad, and a medical-grade adhesive backing, ensuring clear imaging visibility, comfort, and secure fixation.

| Feature | Standard Grid | Metal Grid | No Guide Aid |
|---|---|---|---|
| Risk Level | Low | Medium | High |
| Admission Time | 5-7 days | 7-10 days | 12-14 days |
| Wound Size | Small | Small | Open Surgery |
| Precision | 0.5cm³ | 2cm³ | 10cm³ |
| Imaging | 3D CT Imaging | 2D CT Imaging | Eye Vision |
| Specifications | Packaging | Box Quantity | Equipment |
|---|---|---|---|
| Non-woven 6*9cm | 1pcs/bag | 200pcs/box | DR/CT/DSA/MRI |
| Non-woven 9*9cm | 1pcs/bag | 200pcs/box | DR/CT/DSA/MRI |
| Non-woven 15*15cm | 1pcs/bag | 200pcs/box | DR/CT/DSA/MRI |
| Non-woven 16*16cm | 1pcs/bag | 200pcs/box | DR/CT/DSA/MRI |






As a professional manufacturer of medical consumables, we provide one-stop solutions with a decade of R&D expertise. Our facilities include purification workshops and a massive daily production capacity to meet global demand. We specialize in surgical drapes, wound dressings, and X-ray equipment accessories.
We support full OEM & ODM services, including customized grid formats, sizes, and medical-grade packaging to help our partners grow their clinical portfolio.
