Basic Information
| Model NO. |
CT Grid 001 |
Sterilization |
Ethylene Oxide (EO) |
| Guarantee Period |
Two Years |
Instrument Class |
Class I |
| Application |
Biopsy Positioning Support |
Safety Standard |
EN 149 -2001+A1-2009 |
| Certification |
CE, ISO |
Single Package Size |
14*14cm |
Product Description
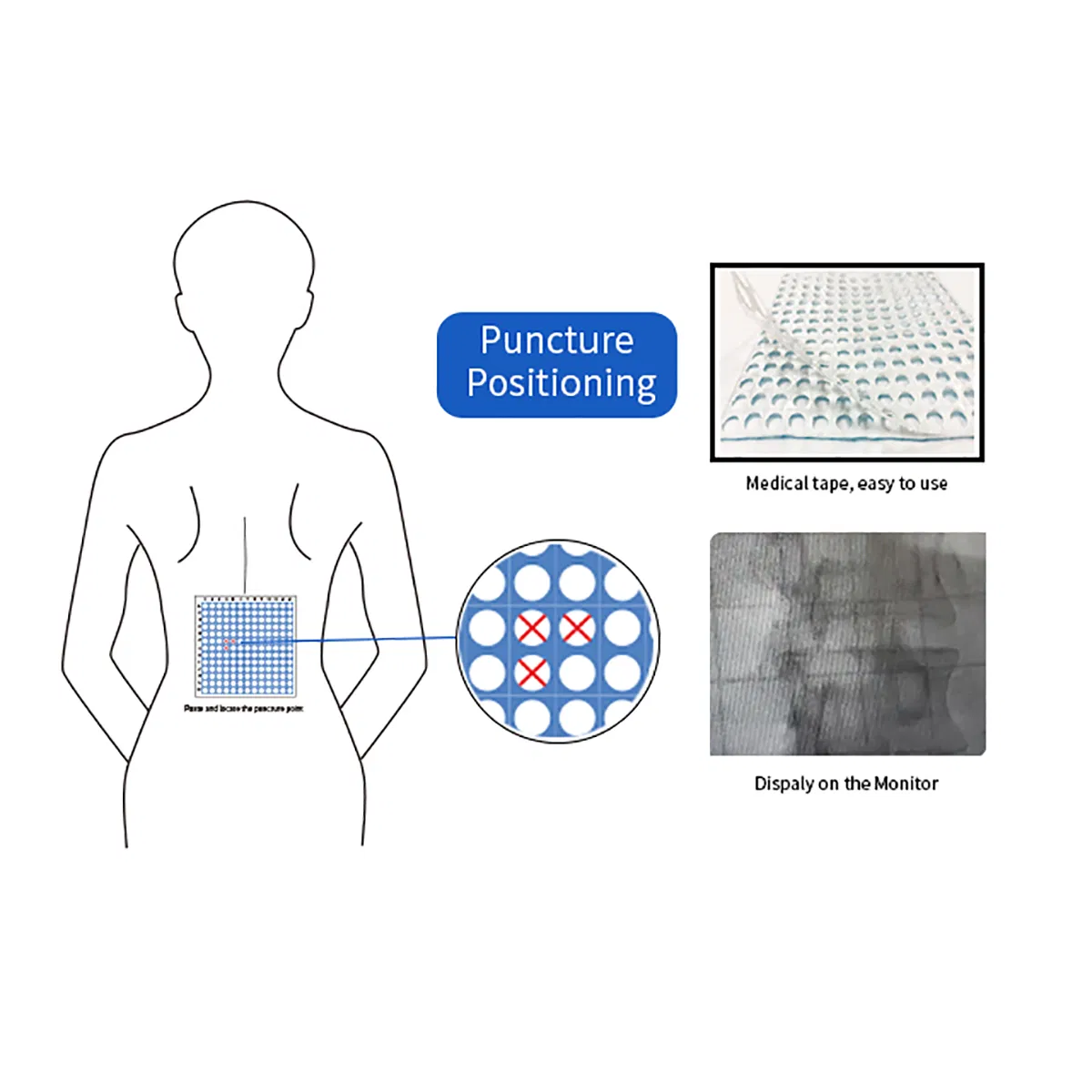
The CT Biopsy Grid is a disposable radiopaque localization grid designed to improve accuracy in image-guided biopsy and drainage procedures. Compatible with CT, DR, DSA, R/F, Ultrasound, and MRI systems, it enables precise alignment between skin markings and imaging coordinates, helping reduce positioning errors, repeated needle adjustments, unnecessary rescans, and patient discomfort.
Featuring a full-surface medical-grade adhesive backing, the grid conforms naturally to different body contours and remains flat and stable on the skin throughout imaging. Constructed with a proprietary three-layer design—patented lead-free radiopaque ink, non-woven 100% medical cotton pad, and medical-grade adhesive—it ensures clear visibility and patient safety.
Why Choose Our Products
- Multi-Modality Compatibility: Suitable for CT, DR, DSA, MRI, US, and Radiographic Fluoroscopy.
- Accurate Localization: 1 cm grid spacing provides precise reference points for needle placement.
- Secure Fixation: Full medical adhesive backing prevents movement during scanning.
- Infection Control: Single-use design minimizes cross-contamination risks.
- Flexible Material: Conforms easily to curved anatomical surfaces.
Comparative Summary & Specifications
| Feature |
Standard Grid |
Metal Grid |
No Guide Aid |
| Risk Level |
Low |
Medium |
High |
| Wound Size |
Small |
Small |
Open Surgery |
| Precision |
0.5cm³ |
2cm³ |
10cm³ |
| Learning Curve |
8-10 cases |
35 cases/year |
35 cases/year |
| Mesh Type Size |
Packaging |
Box Quantity |
Equipment |
| 6*9cm |
1pcs/bag |
200pcs/box |
DR/CT/DSA/MRI |
| 9*9cm |
1pcs/bag |
200pcs/box |
DR/CT/DSA/MRI |
| 15*15cm |
1pcs/bag |
200pcs/box |
DR/CT/DSA/MRI |
| 16*16cm |
1pcs/bag |
200pcs/box |
DR/CT/DSA/MRI |
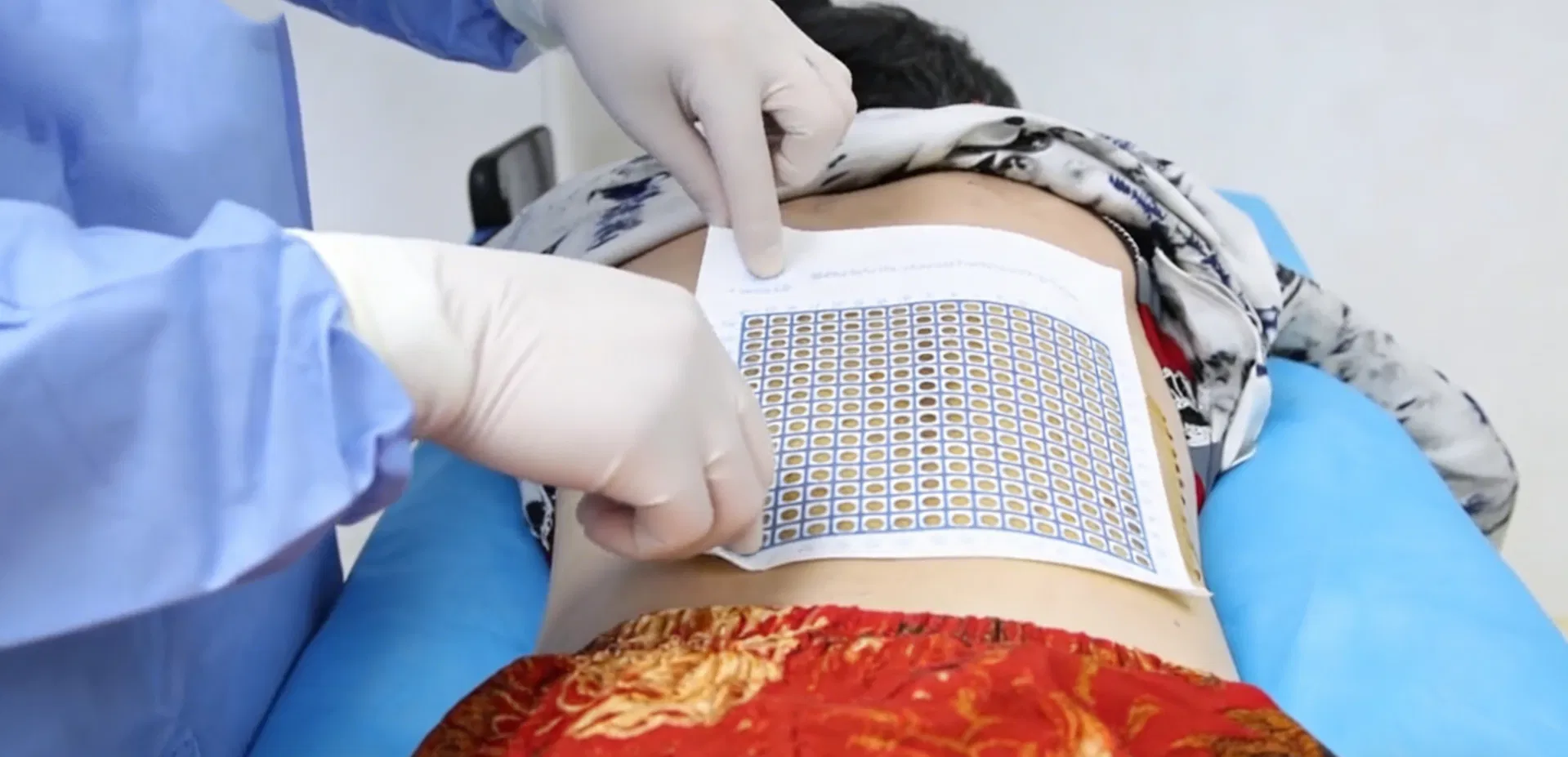
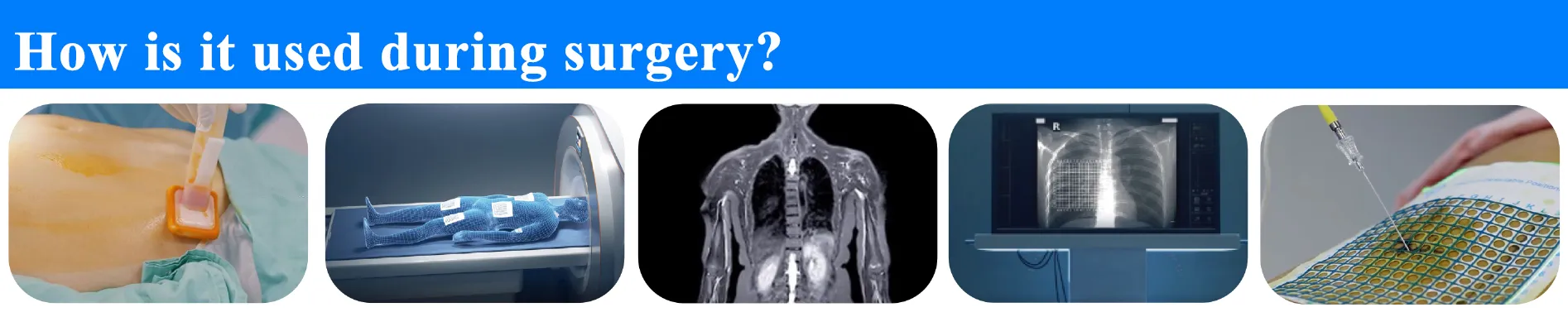
How to Use
1. Disinfect the skin surrounding the application site.
2. Place the product adhesive side down over the area to be imaged.
3. Perform imaging (CT/MRI/DSA) of the target area.
4. Cross-reference scan image with measurement to identify the surface position.
5. Mark the position with a surgical marker and begin the procedure.
Clinical Applications
Key Departments: General Surgery, Interventional Radiology, Pulmonology, Endocrinology, Orthopedics, Oncology, and Emergency Care.
It is used for abdominal/pelvic biopsies, lung biopsy localization, spinal puncture, thyroid nodule puncture, and emergency drainage procedures.
Production Workflow
- Raw Materials: Hypoallergenic medical pressure-sensitive adhesives.
- Precision Cutting: Standardized or customized sizing with no wrinkles or impurities.
- Grid Printing: Clear X-Y axis markings for high-accuracy localization.
- Sterilization: EO or Gamma sterilization as per requirement.
- Quality Control: 100% inspection for dimensional accuracy and CT visibility.
Professional Manufacturing
As a professional medical consumable solution supplier with a 10-year expert R&D team, our company operates three 100,000-class purification workshops. We focus on surgical dressings, ultrasound covers, and X-ray equipment accessories. With CE (MDR), ISO, and FDA certifications, we support global healthcare providers with high-quality, reliable medical tools.
Frequently Asked Questions
Q1: Are you a manufacturer?
A: Yes, we are an integrated industry and trade company specializing in medical consumables for over 25 years.
Q2: Can I get samples for quality testing?
A: Sure, we are glad to provide samples for your evaluation.
Q3: Do you accept small orders?
A: Yes, we accept small orders and are willing to grow with our partners for long-term relationships.
Q4: What certifications do your products have?
A: Our products carry CE and ISO certifications, ensuring compliance with international medical standards.
Q5: Can you offer customized services?
A: Yes, we support both OEM and ODM services, including customization for product specifications and packaging logos.
Q6: What is the typical lead time?
A: Usually, the lead time is between 15-20 days, depending on order volume.




 Aert Medical
Aert Medical